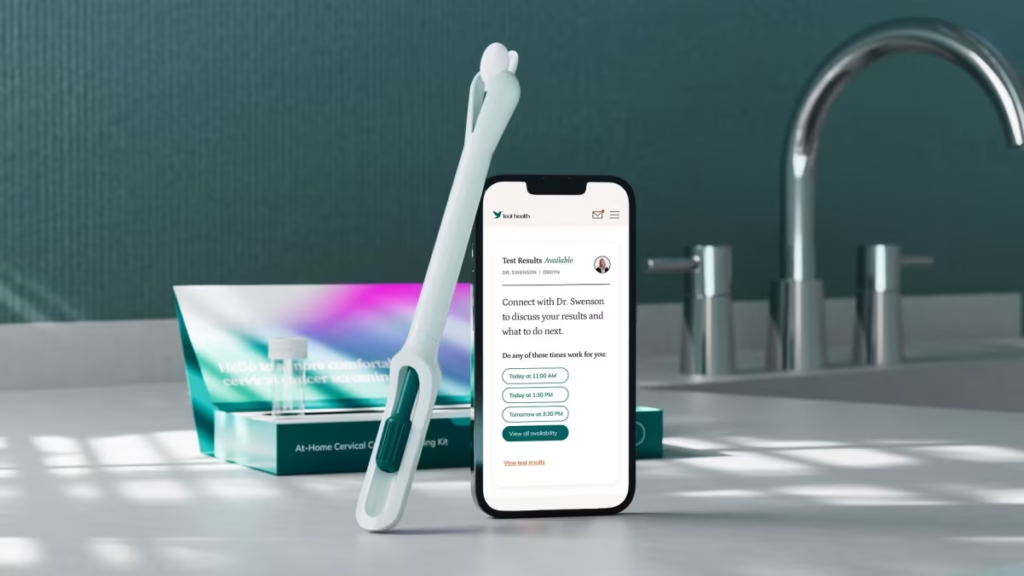
The HPV home screening test has been approved by the U.S. Food and Drug Administration. This marks a major step in women’s health care. It allows people to test for human papillomavirus, also known as HPV, at home. HPV is the main cause of cervical cancer.
Health experts say this move may change how screening is done. Many women avoid clinic visits due to time, cost, or fear. The new system aims to remove these barriers. It makes testing simpler and more private.
Cervical cancer develops slowly in most cases. It often starts with HPV infection. If found early, it can be treated or even prevented. Regular screening has already reduced death rates in many countries. But many people still miss tests.
The HPV home screening test helps solve this problem. Women can now collect a sample at home. The sample is then sent to a lab for analysis. Results are shared later through secure systems. This process is simple and less stressful for many users.
The approval comes after years of research and clinical testing. Health authorities reviewed data on accuracy and safety. They found the home test to be reliable for HPV detection. This is important for early warning signs of cervical cancer risk.
Doctors say early detection is key. When HPV is found early, treatment can start sooner. This reduces the chance of cancer developing. It also lowers long-term health costs for patients and health systems.
Experts believe the HPV home screening test will help reach more people. Rural areas and low-income groups often face limited access to clinics. Home testing can help close this gap. It can also support women who feel uncomfortable with in-clinic exams.
Public health groups also support the decision. They say screening rates must rise to reduce cervical cancer deaths. Many countries still report low testing coverage. Easy home access may improve these numbers over time.
The test is designed for safety and ease of use. Clear instructions are provided. Users collect a sample using a simple kit. The kit is then sealed and mailed to a certified lab. No special training is needed.
Medical professionals still stress the need for follow-up care. A home test does not replace doctors. It is a first step in screening. If results show risk, further medical checks are required.
Some experts also highlight data privacy. They say strong systems must protect patient results. Health agencies say the approved system includes secure handling of all data.
The HPV home screening test may also reduce pressure on clinics. Many health systems face long waiting times for screening appointments. Home testing can help reduce this load. It allows clinics to focus on high-risk patients.
Global health researchers are watching this development closely. If successful, other countries may adopt similar systems. This could lead to wider use of home-based screening tools in the future.
Cervical cancer remains a serious health issue worldwide. But it is also one of the most preventable cancers. Vaccines and regular screening already help reduce risk. The new home test adds another layer of protection.
The FDA approval reflects growing trust in at-home medical technology. More health tools are moving outside hospitals and into homes. This trend may continue as technology improves.
For now, health experts encourage eligible women to stay informed. Regular screening remains very important. The HPV home screening test gives a new, easier option for many people.